Lithium-Ion Batteries
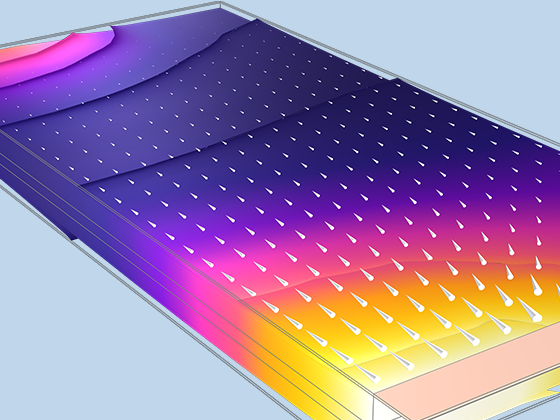
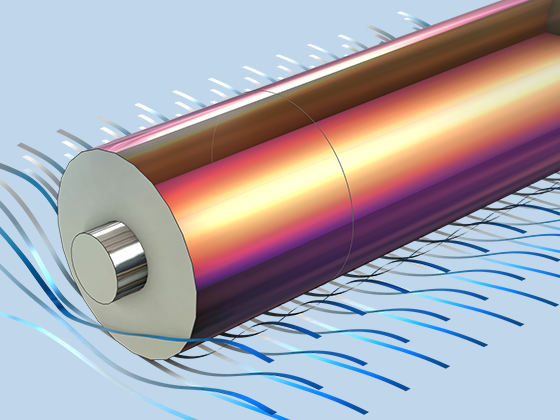
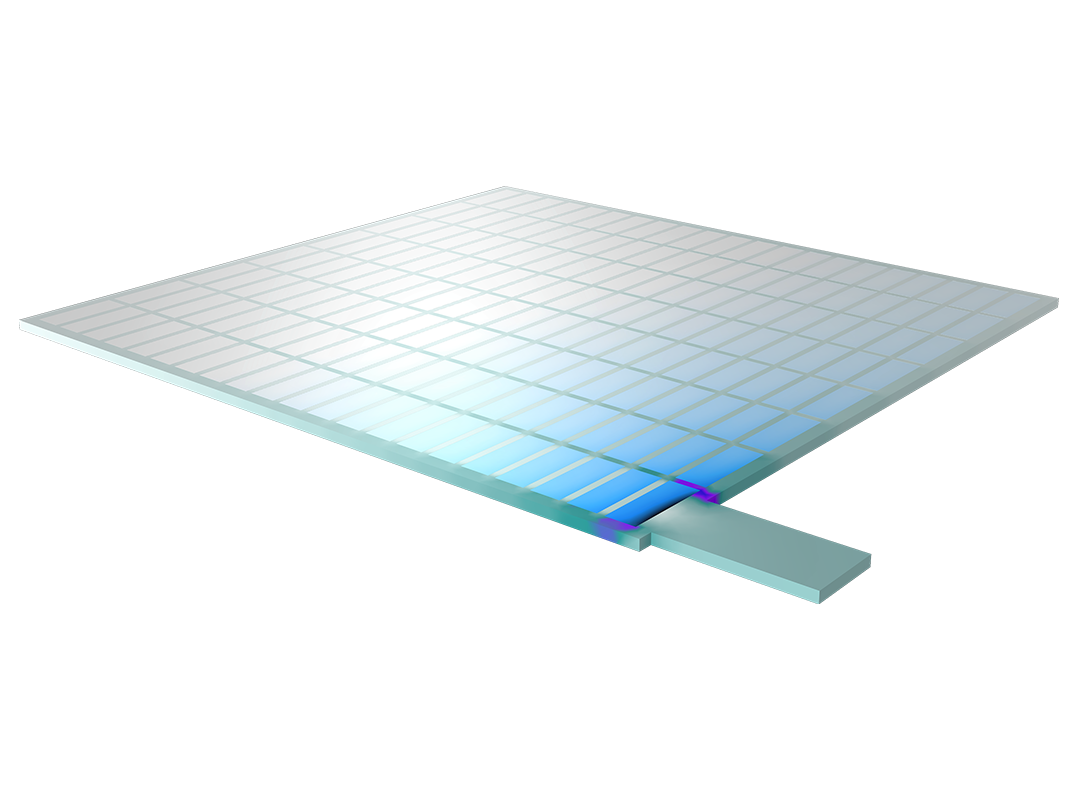
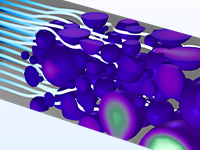
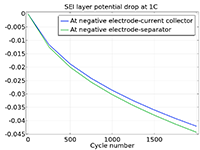
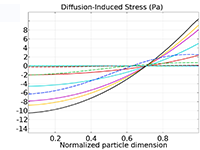
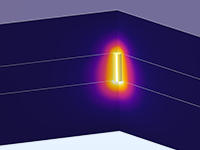
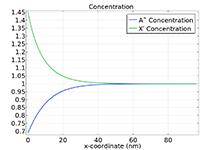
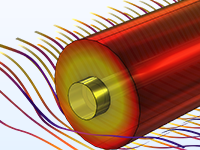
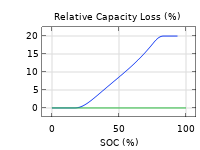
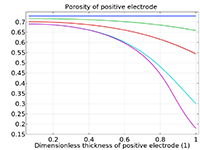
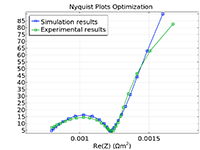
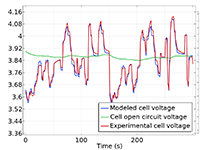
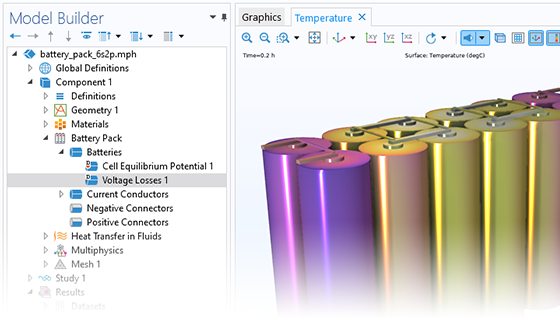
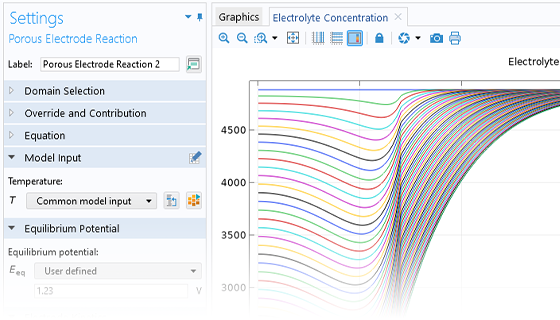
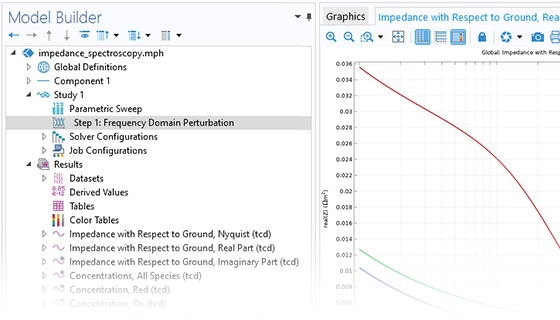
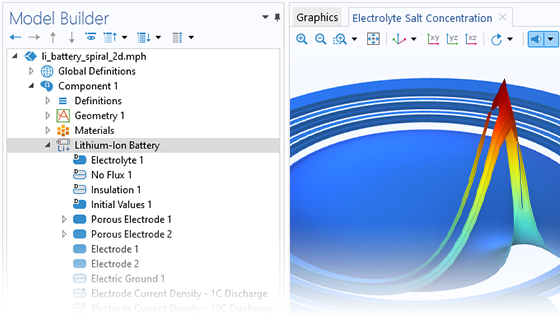
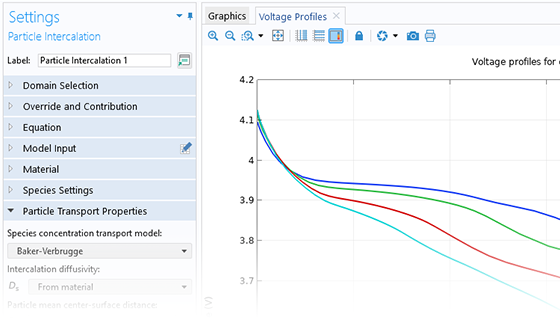
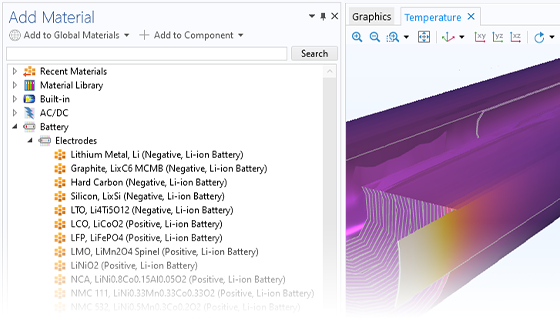
The Battery Design Module features state-of-the-art models for lithium-ion batteries. You will find different mechanisms for aging and high-fidelity models, such as the Newman model, available in 1D, 2D, and full 3D. In addition to modeling electrochemical reactions on their own, you can combine them with heat transfer and account for the structural stresses and strains caused by the expansion and contraction from lithium intercalation. The module also provides functionality for setting up heterogenous models, which describe the actual shapes of the pore electrolyte and electrode particles. Studying the microstructure of a battery helps to provide a deeper understanding of the battery performance.